Table of Contents
Source of water: Burning of candle wax in air
Reactions of Water with Metals
Reactions of Metals with Steam
Laboratory Preparation and properties of Hydrogen
Past KCSE Questions on Water and Hydrogen
Objectives of this Lesson (Water and Hydrogen)
By the end of this chapter, the learner should be able to:
- Describe experiments to show that water is a product of burning organic matter and that it contains hydrogen.
- State the products of reactions of metals with cold water and steam then derive the reactivity series of the metals.
- Prepare hydrogen and investigate its properties and uses.
(20 Lessons)
WATER AND HYDROGEN
Water
Water is the most abundant substance on earth. About 71% of the earth’s surface is covered by water. The sources of water are seas, lakes, rivers and oceans.
Source of water: Burning of candle wax in air
Carbon and hydrogen are the major components of organic matter. When organic matter burns in air, carbon (IV) oxide and water are produced. Candle wax is made up of organic matter.
Candle wax is a compound of carbon and hydrogen. It belongs to a group of compounds called hydrocarbons. It burns in air to form carbon (IV) oxide and water.
Other hydrocarbons include kerosene, petrol and diesel.
⚗Practically Speaking🔊📌
To demonstrate what happens when the candle wax burns in the air, the set-up below can be used.
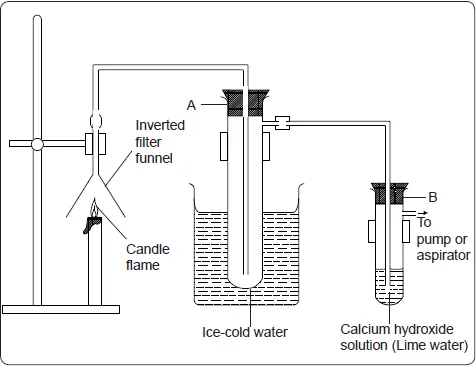
Light the candle and turn on the pump. Allow the candle to burn for about 15 minutes. Observe and record what happens in tubes A and B.
Divide the contents of test tube A into two portions. To one portion, add anhydrous copper (II) sulphate or blue cobalt (II) chloride paper. Record your observations.
Side notes
- Test-tube A is dipped in cold water so that the cold water condenses the vapour to a liquid.
Observations and Discussion
In test-tube A, a colourless liquid, which turns white anhydrous copper (II) sulphate to blue and blue cobalt (II) chloride paper to pink is observed.
In test tube B, a white precipitate is observed.
Candle wax burns in the air to form a colourless liquid, which turns white anhydrous copper(II) sulphate to blue and blue cobalt (II) chloride paper to pink. The liquid formed is water. A colourless gas that forms a white precipitate with lime water is also produced. This gas is carbon (IV) oxide.
Reactions of Water with Metals
Water reacts with some metals to produce hydrogen gas.
⚗Practically Speaking🔊 📌
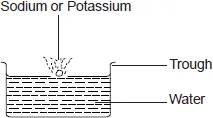 The following experiments can be done to showcase the reaction between water and some metals
The following experiments can be done to showcase the reaction between water and some metals - Cut a very small piece of sodium metal. Drop it into a trough containing cold water. Test the solution in the trough using pieces of red and blue litmus paper. Record your observations.
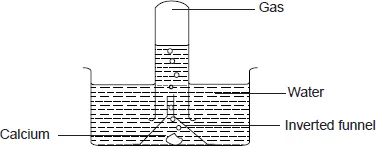
- Drop a piece of calcium metal into water in a trough. Invert a funnel over the calcium. Fill a test tube with water and invert it over the funnel. Collect the gas given off. Cork the test-tube to prevent the gas from escaping. Lift the test-tube from water and turn it upright. As you remove the cork, bring a burning splint near the mouth of the test-tube. Observe what happens. Test the solution in the beaker using pieces of red and blue litmus paper. Record your observations.
- Put a two-centimetre piece of clean magnesium ribbon into a test-tube containing water. Record your observations. Repeat the experiment using clean zinc granules and fresh iron filings respectively.
Side notes
- It is necessary to clean the magnesium ribbon before using because magnesium reacts with atmospheric oxygen to form a coating of magnesium oxide. The coating has to be removed so that the metal surface comes into contact with the water.
- The magnesium ribbon is coiled in order to increase the surface area in contact with the boiling tube.
Observations and Discourse
When a piece of sodium metal is placed in water, it melts into a silvery ball as it reacts vigorously darting on the surface of the water with a hissing sound. The resulting solution turns red litmus blue showing that it is basic.
Sodium + Water
When calcium is added to water it sinks and reacts moderately with water producing a steady stream of bubbles. When the gas is tested with a burning splint, it burns with a ‘pop’ sound indicating the gas is hydrogen. A basic solution of calcium hydroxide is formed.
A white suspension is observed because the calcium hydroxide is slightly soluble in water.
Calcium + Water ![]() Calcium hydroxide + Hydrogen
Calcium hydroxide + Hydrogen
Magnesium reacts very slowly with cold water while zinc and iron do not react.
The reaction of potassium with water is explosive. A small piece of potassium placed on water melts into a silvery ball and moves about rapidly on the surface. The reaction generates a lot of heat. As a result, hydrogen gas produced ignites spontaneously.
The flame produced is lilac (purple) due to the presence of potassium vapour produced during the reaction.
Potassium + Water ![]() Potassium hydroxide + Hydrogen
Potassium hydroxide + Hydrogen
The resulting solution turns red litmus blue showing that it is basic.
Reactions of Metals with Steam
Some metals like magnesium and iron react slowly with cold water. However, with steam, the reaction is faster.
⚗Practically Speaking🔊 📌
The experiment below can be used to demonstrate the reaction between steam and some metals.
Place clean, wet sand in a boiling tube. Clean a piece of magnesium ribbon about 6cm long and make it into a spiral coil. Place it in the middle of the boiling tube and arrange the apparatus as shown below.
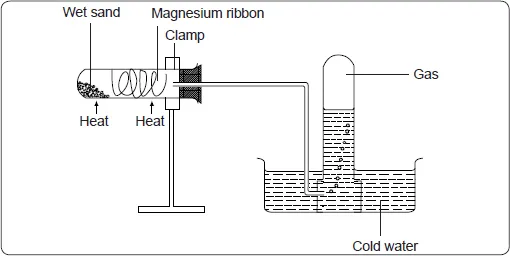 Heat the magnesium strongly and warm the wet sand gently as you continue heating the magnesium. Observe what happens. Remove the delivery tube before you stop heating.
Heat the magnesium strongly and warm the wet sand gently as you continue heating the magnesium. Observe what happens. Remove the delivery tube before you stop heating.
Test the gas produced using a burning splint. Record your observations. Repeat the experiment using zinc powder and iron filings.
Side notes
- The wet sand is warmed to generate steam, which reacts with the metal.
- The delivery tube is removed before heating stops to prevent water being sucked into the hot boiling tube as it cools.
Observations and Discussion
Magnesium burns brightly in steam to form white magnesium oxide powder and hydrogen gas.
Magnesium + Steam ![]() Magnesium oxide + Hydrogen
Magnesium oxide + Hydrogen
Zinc and iron do not burn in steam, they glow. Zinc metal reacts with steam to form a yellow powder of zinc oxide, which turns white on cooling.
Zinc + Steam ![]() Zinc oxide + Hydrogen gas
Zinc oxide + Hydrogen gas
Iron forms a black residue of an oxide called tri-iron tetraoxide and hydrogen.
Iron + Steam ![]() Tri-iron tetra oxide + Hydrogen gas
Tri-iron tetra oxide + Hydrogen gas
Aluminium reacts with steam but quickly forms a layer of aluminium oxide which prevents further reaction. Lead and copper do not react with either cold water or steam.
Summary of reaction of metals with cold water and steam | ||
Metal | Action of metal on water | Action of metal on steam |
| Potassium | Violent | Explosive |
| Sodium | Violent | Explosive |
| Calcium | Moderate | Violent |
| Magnesium | Very slow | Rapid |
| Aluminium | No reaction | Slow |
| Zinc | No reaction | Slow |
| Iron | No reaction | Slow |
| Lead | No reaction | No reaction |
| Copper | No reaction | No reaction |
Metals can be arranged in order of their reactivity with water. The arrangement gives the Reactivity Series of metals, as shown below.
Potassium Most reactive
Sodium
Lithium
Calcium
Magnesium
Aluminium
Zinc
Iron
Lead
Copper Least reactive
Hydrogen
Hydrogen is the simplest and lightest element. It does not occur as a free element on earth but exists in a combined form. Examples of compounds containing hydrogen are water, hydrocarbons and sugar.
Laboratory Preparation and properties of Hydrogen
Hydrogen gas is prepared in the laboratory by the reaction between dilute acids and some metals. Dilute sulphuric acid and hydrochloric acid react with some metals to liberate hydrogen gas. Zinc is the most suitable metal for the laboratory preparation of hydrogen gas.
⚗Practically Speaking🔊 📌
The set up below can be used to prepare hydrogen gas in the laboratory.
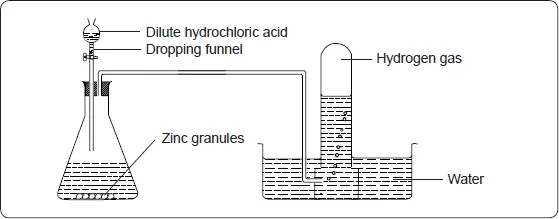
If the reaction is slow, add a few crystals of copper (II) sulphate. If the gas is required dry, it should be bubbled through concentrated sulphuric acid and collected by upward delivery.
Side notes
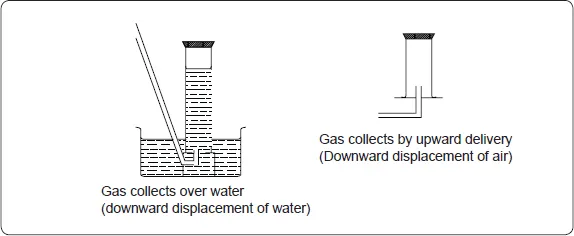
- Hydrogen is insoluble in water, so it is collected over water.
- Hydrogen is less dense than air and it can also be collected by upward delivery. This method is also called downward displacement of air.
- Copper (II) sulphate acts as a catalyst to speed up the reaction.
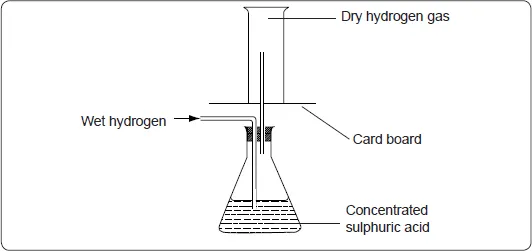
- Dry hydrogen gas can be obtained by passing wet hydrogen through anhydrous calcium chloride in a U-tube or passing it through concentrated sulphuric acid.
Observations and Discussion
When zinc granules are added to dilute hydrochloric acid there is effervescence and hydrogen gas is evolved.
Zinc + Hydrochloric acid ![]() Zinc chloride + Hydrogen
Zinc chloride + Hydrogen
Nitric acid is not used to prepare hydrogen gas because the hydrogen formed is oxidised to water. However, very dilute nitric acid liberates hydrogen with magnesium metal.
Zinc is preferably used because it reacts moderately with dilute acids.
Potassium, sodium, lithium and calcium react explosively with dilute acids hence must not be used. Magnesium could be used for the laboratory preparation of hydrogen but it is expensive. Aluminium forms a protective layer of aluminium oxide, which should be washed using concentrated hydrochloric acid before the metal can react with dilute acids. Impure iron gives a mixture of gases including the bad-smelling hydrogen sulphide when it reacts with dilute acids.
Physical Properties of Hydrogen
Hydrogen is a colourless, odourless gas.
It is less dense than air and insoluble in water.
Hydrogen has no effect on litmus paper. It is neutral
Test for hydrogen
When mixed with air, hydrogen burns with a ‘pop’ sound. This is the test for hydrogen gas. The intensity of the pop sound diminishes as the purity of hydrogen increases. Pure hydrogen burns quietly with a blue flame. Hydrogen does not support combustion but it burns in air.
Reducing property of hydrogen
Hydrogen is a strong reducing agent. It can reduce some metal oxides to their respective metals and is itself oxidised to water.
⚗Practically Speaking🔊 📌
The experiment below can be done to demonstrate the reducing properties of hydrogen.
Place a spatula of copper (II) oxide in a porcelain boat or on a piece of aluminium foil. Place the boat or aluminium foil in a combustion tube. Set the apparatus as shown.
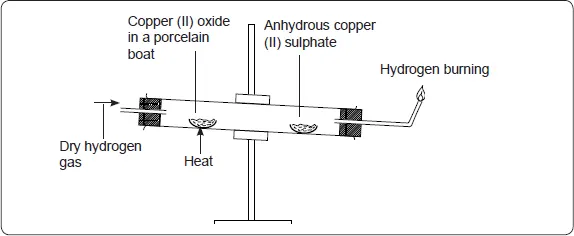
Pass dry hydrogen through the tube for some time to drive out the air from the apparatus. Collect a sample of the gas from the jet in a test-tube and test it with a burning splint. Continue collecting and testing until a sample of the gas burns silently (without a ‘pop’ sound).
Light the gas at the jet and start heating copper (II) oxide as shown. Ensure a steady flow of hydrogen gas. Continue heating until there is no further change. Observe what happens. Allow the apparatus to cool as you continue passing the stream of hydrogen gas over the residue. Disconnect the apparatus and test the colourless liquid in the cooler parts of the combustion tube.
Side notes: Precautions before and after the experiment
- Air is driven out of the apparatus before heating the copper (II) oxide to ensure the hydrogen being burned at the jet is pure to avoid explosion.
- Excess hydrogen gas is burned because its mixture with oxygen (air) is explosive when ignited. The excess gas is therefore not allowed to escape into the air for safety reasons.
- The supply of hydrogen gas is continued while the apparatus cools to avoid the re-oxidation of the hot metal by oxygen from the air.
Observations and Discussion
- On passing a stream of dry hydrogen gas over hot copper (II) oxide the black copper (II) oxide changes to red-brown. At the same time, a colourless liquid condenses and collects on the cooler parts of the combustion tube.
Hydrogen combines with oxygen from the copper (II) oxide to form water which is a colourless liquid. The red-brown solid is copper metal.
Copper (II) oxide + Hydrogen ![]() Copper metal + Water
Copper metal + Water
In this reaction copper (II) oxide is reduced to copper metal while hydrogen is oxidised to water.
Hydrogen removes oxygen from copper (II) oxide and is therefore referred to as a reducing agent. Copper (II) oxide loses oxygen to hydrogen hence it is an oxidising agent. Hydrogen will also reduce the oxides of lead and iron.
The removal of oxygen from a compound is known as reduction while the addition of oxygen is known as oxidation.
Lead (II) oxide + Hydrogen ![]() Lead metal + Water
Lead metal + Water
Iron (III) oxide + Hydrogen ![]() Iron metal + Water
Iron metal + Water
Hydrogen does not remove oxygen from oxides of more reactive metals.
Burning of hydrogen in air
Hydrogen combines with oxygen from the air to form water. Water is therefore an oxide of hydrogen.
⚗Practically Speaking🔊 📌
To demonstrate the burning of hydrogen in air, arrange the apparatus as shown below.
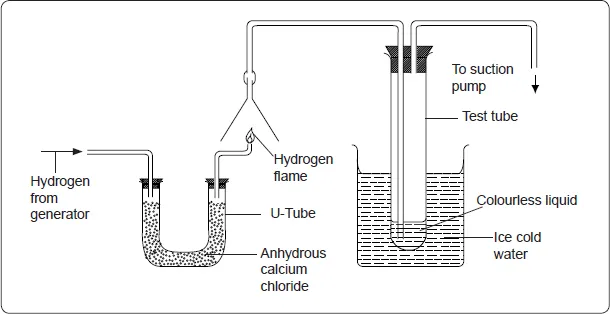
Pass a stream of hydrogen gas through anhydrous calcium chloride. Test the gas for purity by collecting samples over the jet and testing with a burning splint until the gas burns silently. Light the gas at the jet and turn on the pump. Draw in the products of the burning hydrogen using the pump for about 15 minutes. Test the liquid in the test tube using white anhydrous copper (II) sulphate.
Side notes
- Anhydrous calcium chloride is used to dry the gas.
- The ice-cold water condenses the steam to form liquid water.
Discourse
When hydrogen burns in air a colourless liquid which turns white anhydrous copper (II) sulphate blue is formed. The liquid is water.
Hydrogen + Oxygen ![]() Hydrogen oxide (water)
Hydrogen oxide (water)
Uses of Hydrogen
1. Hydrogen is used in the large-scale manufacture of ammonia in a process known as the Haber process.
2. The gas is also used during the hardening of oils to form fats. When hydrogen is passed through liquid oil in the presence of a nickel catalyst, the oil takes up hydrogen and is converted into fat. This process is called hydrogenation. It is used in the manufacture of margarine.
3. Hydrogen is used in balloons because it is lighter than air. A balloon filled with hydrogen floats in air. A light radio instrument can be connected to the balloon to collect information from the atmosphere by meteorologists who study weather conditions.
4. A mixture of hydrogen and oxygen burns to produce a very hot flame, the oxy-hydrogen flame, which has a temperature of up to 2000°C. The flame is used in welding and for cutting metals.
5. Hydrogen is used in rockets as fuel.
6. Hydrogen is used in the manufacture of hydrochloric acid.
7. Hydrogen is used as a fuel in fuel cells.
Review Exercises on Water and Hydrogen
- Name two substances formed when a candle burns in the air.
- The following set-up was used to investigate the products formed when a candle burns.
- (a) You are provided with a colourless odourless liquid which you suspect to be pure water State how you would test for the purity of this liquid to confirm it is pure water.
- ((a) Draw the set-up of apparatus you would use to burn dry hydrogen gas and collect the product.
- (a) Write a word equation for the reaction that takes place when dry hydrogen gas is passed over heated copper (II) oxide in a combustion tube.
- From the list of metals: lead, magnesium, iron, potassium, calcium, sodium and zinc, name the metal or metals that:
- In an experiment, steam was passed over heated zinc metal using the following set-up of apparatus.
- State three physical properties and three chemical properties of hydrogen gas.
- A student arranged apparatus as shown below to prepare and collect dry hydrogen gas.
- Steam is passed over heated magnesium as shown in the diagram below.
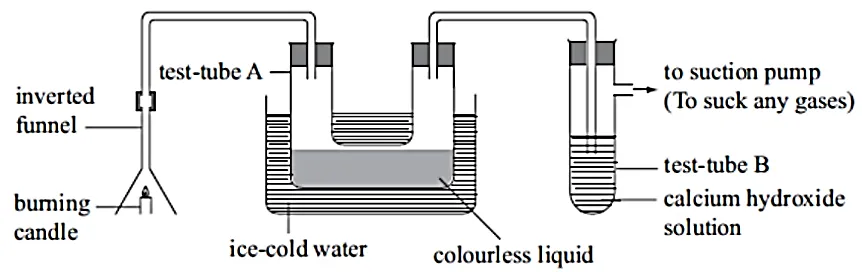
(a) State the observations made in
(i) Test tube A.______________
(ii) Test tube B_____________
(b) Explain the observations you stated in (a) (i) and (ii) above.
(c) State the effect of the contents of test tube A on anhydrous cobalt chloride paper
(d) Give one physical test for the contents of test tube A.
(e) Name two products formed when the candle burns.
(f) What elements are present in candle wax?
b) A student left dry cobalt (II) chloride paper exposed overnight. The colour of the paper changed from blue to pink. Explain.
(b) Write a word equation for the combustion of hydrogen.
(c) What type of reaction does hydrogen undergo during combustion?
(d) State the precaution you would take at the start and end of this combustion reaction
(e) State any four uses of hydrogen.
(b) State the observations you would make in the combustion tube.
(a) react at a moderate rate with cold water;
(b) react with steam only when strongly heated;
(c) does not react with water or steam.
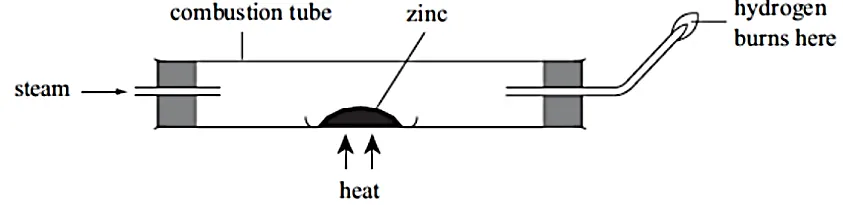
(b) Write a word equation for the reaction that occurs in the combustion tube.
(c) Write a word equation for the reaction that produces the flame.
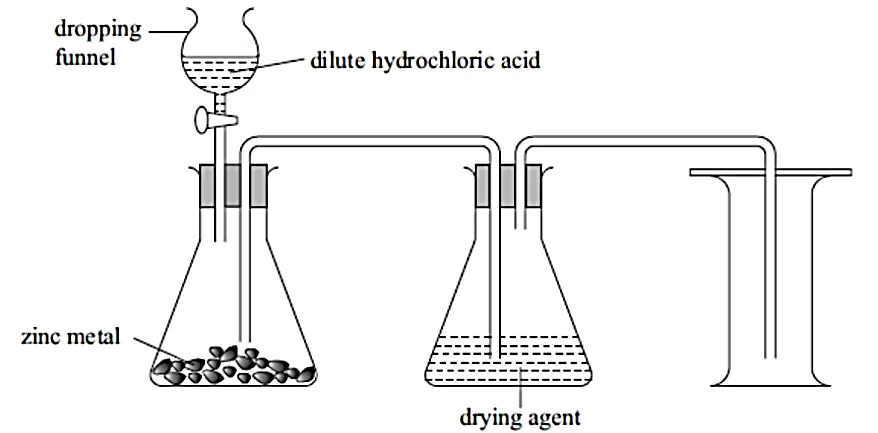
(a) Identify two mistakes in this setup.
(b) If the dropping funnel is replaced with a thistle funnel, state the correction that should be made in the set-up.
(c) Name one metal that cannot be used in this experiment. Explain.
(d) Name a suitable drying agent that can be used in this experiment.
(e) Write a word equation for the reaction that takes place.
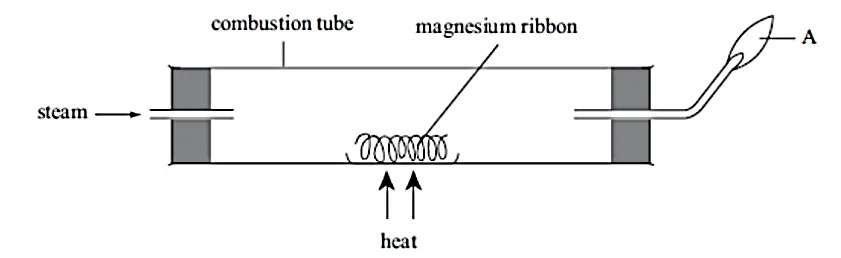
(a) Give two observations that will be made in the combustion tube during the experiment.
(b) Name the substance that was
(i) Burning at A
(ii) Produced at A
(c) Write a word equation for the reaction in the combustion tube.
(d) Compare the initial mass of magnesium ribbon with that of the product. Explain.
Past KCSE Questions on Water and Hydrogen
- 2006 Q 18 P1
- 2006 Q 19 P1
- 2007 Q 25a P1
- 2008 Q 13 P1
- 2009 Q 4 P1
- 2009 Q 15 P1
- 2009 Q 5c P2
- 2011 Q 9 P1
- 2011 Q 12 P1
- 2012 Q3 P1
- 2012 Q19 P1
- 2013 Q18 P1
- 2015 Q9 P1, 2016 Q4 P1
- 2017 Q3 (b) P2
- substance X.
ice-cold water Y (1 mark)b) Give the name of the substance that could be used as X. (1 mark)
c) State the role of the suction pump. (1 mark)
d) Name the product Y formed. (1 mark)
e) Give a simple physical test to prove the identity of Y. (1 mark)
f) State the difference between ‘dry’ and ‘anhydrous’. (2 marks)
- 2018 Q 21 P1
- 2019 Q5 P1
Study the diagram below and answer the question that follows.
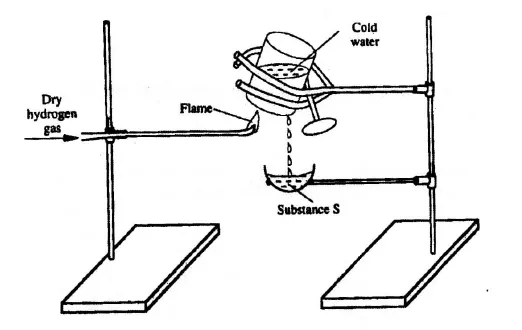
Describe one chemical test that can be carried out to identify substance S. (2 marks)
Starting from solid magnesium hydroxide, describe how a solid sample of magnesium hydroxide can be prepared (2 marks)
Give one use of magnesium hydroxide. (1 mark)
The diagram below shows a student’s set-up for the preparation and collection of hydrogen gas.
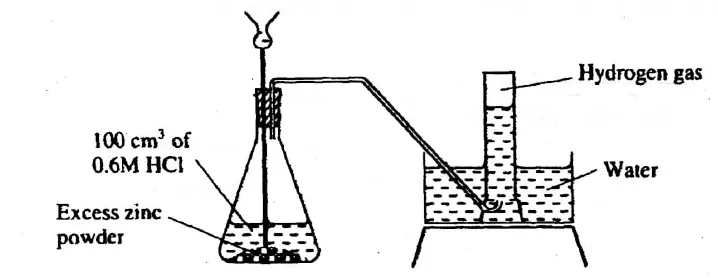
How would the final volume of hydrogen gas produced be affected if 80cm3 of 0.75 M hydrochloric acid was used? (1 mark)
In a laboratory experiment hydrogen gas was passed over heated copper (II) oxide as shown the diagram below.
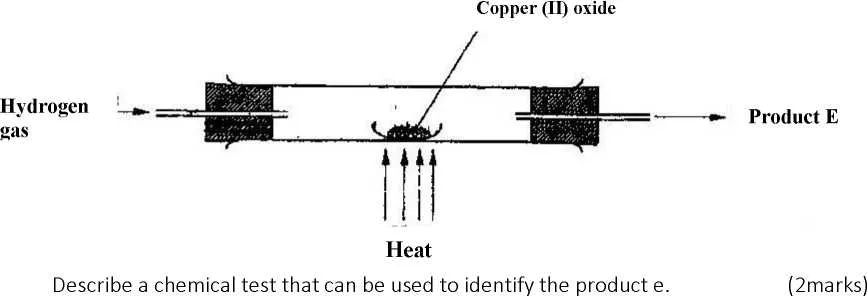
Describe a chemical test that can be used to identify the product E. (2 marks)
An experiment was set up shown in the diagram below.
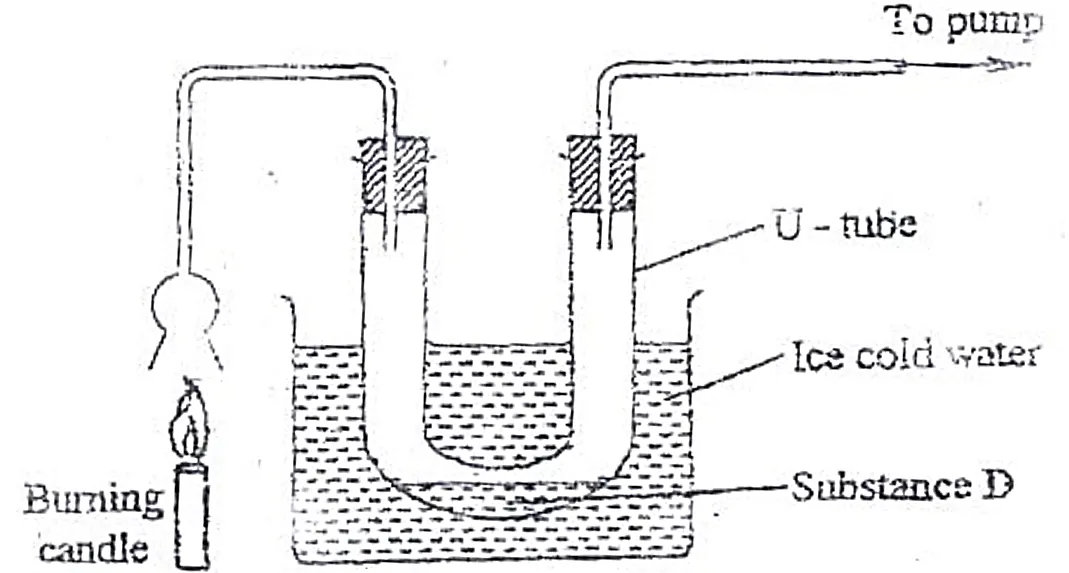
Identify substance D (1 mark)
Describe how the other product of the burning candle could be prevented from getting into the environment. (2 marks)
A student used the set up shown in the diagram below in order to study the reactions of some metals with steam. The experiment was carried out for ten minutes.
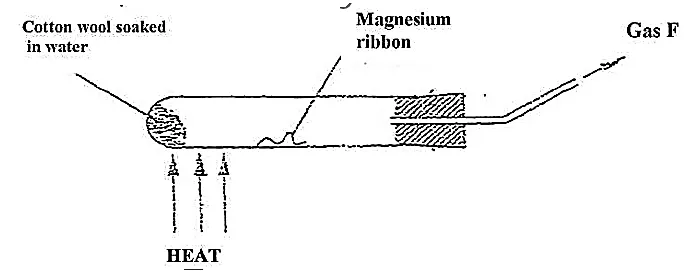
What observation would be made if gas F is ignited? (1 mark)
When the experiment was repeated using iron powder instead of magnesium ribbon, very little gas F was obtained.
Give a reason for this observation (1 mark)
What change in the conditions of the experiment should the student have made in order to increase the volume of gas F produces. (1 mark)
Coal, oil and natural gas are major sources of energy. They are known as fossil Hydrogen is also a source of energy.
State and explain two reasons why hydrogen is a very attractive fuel compared to fossil fuels (3 marks)
State one disadvantage of using hydrogen fuel instead of fossil fuel. (1 mark)
State two reasons why hydrogen is not commonly used as a fuel. (2 marks)
Sodium hydroxide can be prepared by the following methods;I and II.
Sodium metal Sodium hydroxide + Hydrogen
Concentrated Sodium hydroxide + chlorine + hydrogen
Sodium chloride
Name one precaution that needs to be taken in method 1. (1 mark)
Give the name of process A. (1 mark)
Give one use of sodium hydroxide. (1 mark)
In an experiment, dry hydrogen gas was passed over heated Lead (II) Oxide as shown in the diagram below.
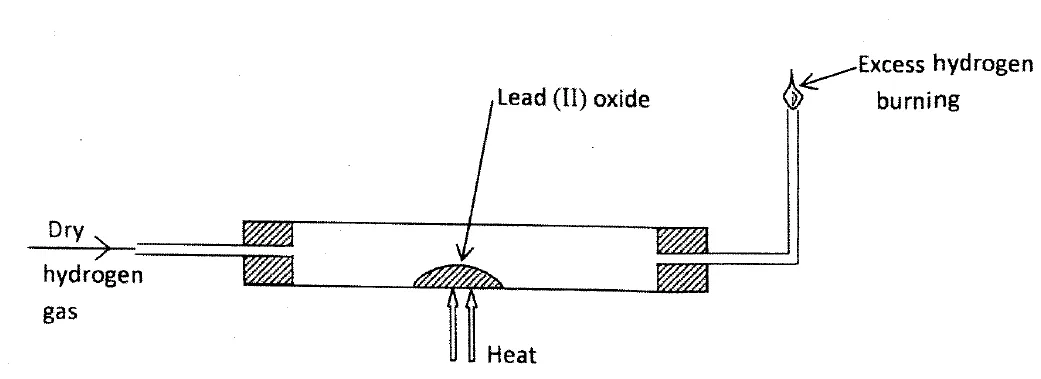
State and explain the observations made in the combustion tube (3 marks)
The set up shown below was used to investigate a property of hydrogen gas.
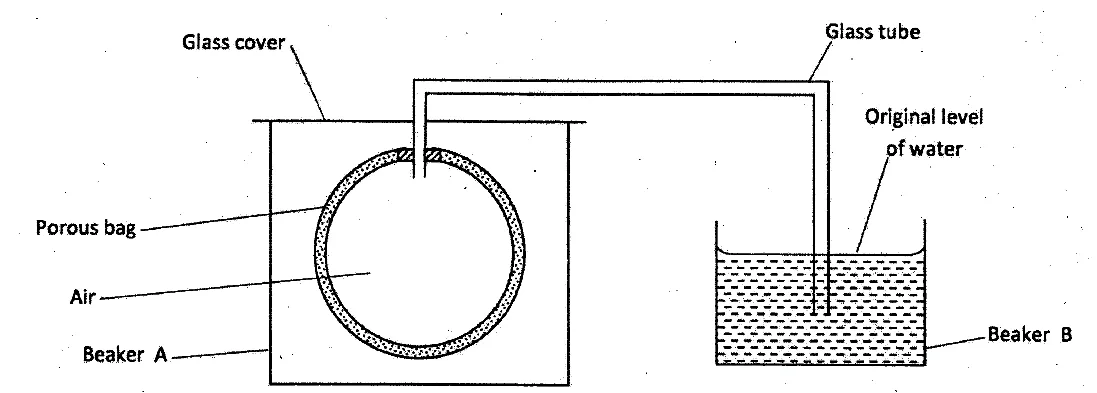
State and explain the observation that would be made in the glass tube if beaker A was filled with hydrogen gas. (3 marks)
The set-up below was used to investigate the products of burning biogas (methane). Study it and answer the questions that follow.
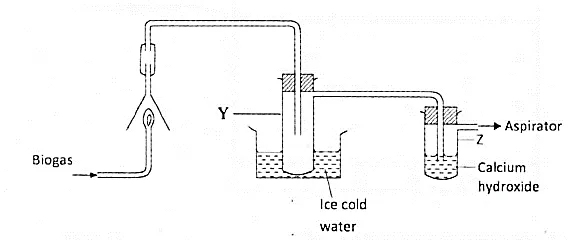
What product will be formed in test- tube Y?
State and explain the observations which would be made in Z?
The set up below was sued to investigate the reaction between dry hydrogen gas and copper (II) oxide
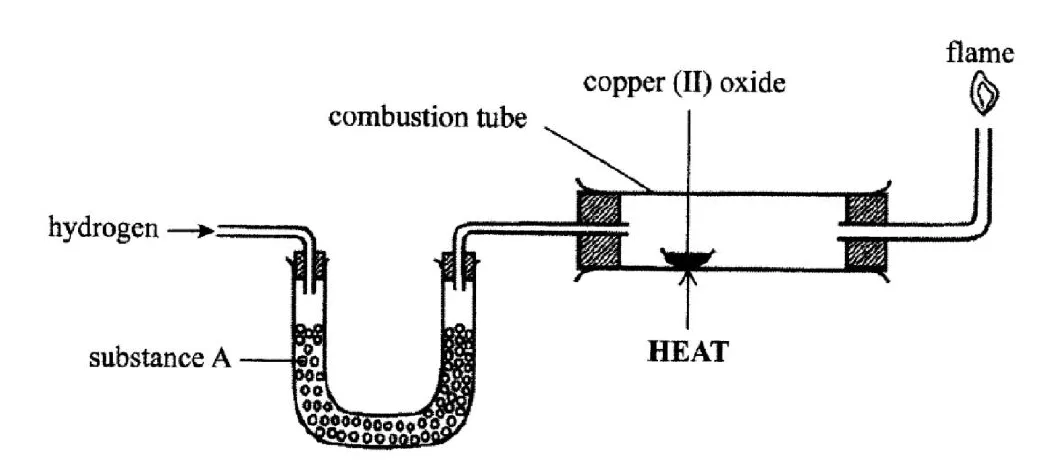
Name substance A. (1 mark)
State the observation made in the combustion tube. (1 mark)
Explain the observation made in (b) above. (1 mark)
Figure 3 shows the apparatus used to burn hydrogen in air. Use it to answer the questions that follow.
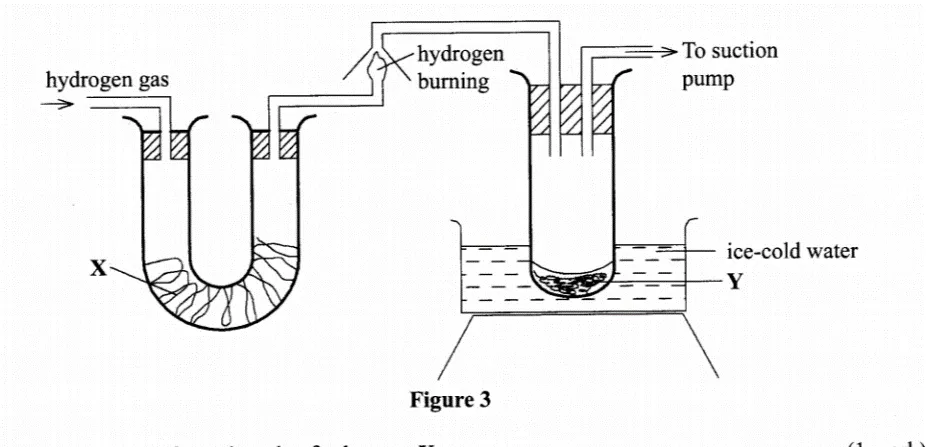
a) State the role of:
Study the set-up in Figure 6 and answer the questions that follow.
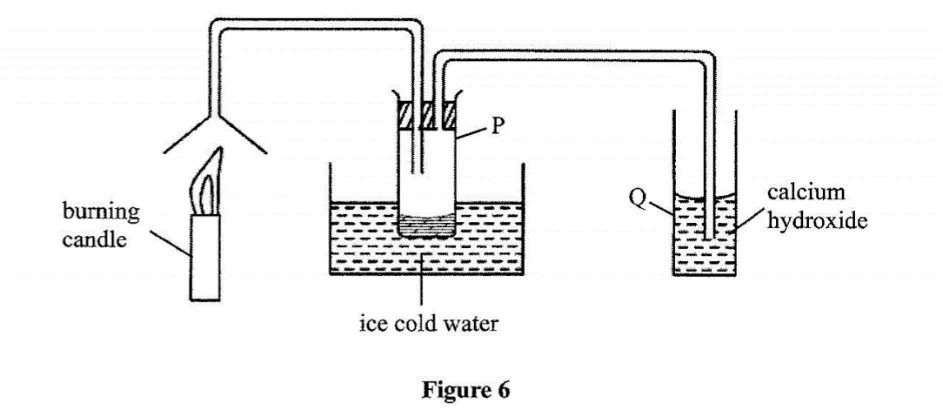
Name the substance that was collected in tube P. (1 mark)
Write an equation for the reaction which occurs in tube Q in the first few minutes of the experiment. (1 mark)
Give a suitable conclusion for the experiment in the set-up. (1 mark)
Hydrogen gas can be prepared by passing steam over a heated magnesium ribbon as shown in the figure 2.
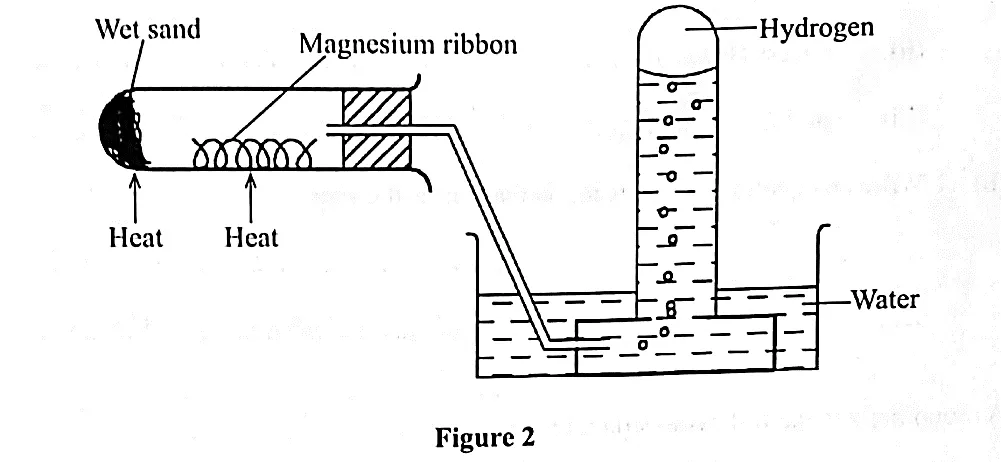
- Write an equation for the reaction that produces hydrogen gas. (1 mark)
Explain why the delivery tube must be removed from beneath the water before heating is stopped. (1 mark)
Explain why sodium metal is not suitable for this experiment. (1 mark)



Comments are closed.