Table of Contents
- Simple classification of substances
- Mixtures
- Separation of solid- solid mixtures
- Solid-liquid mixtures
- Separation of insoluble solid-liquid mixtures
- Separation of soluble solid-liquid mixtures
- Liquid – liquid mixtures
- Separation of immiscible liquids
- Separation of miscible liquids
- Separation of coloured substances
- Effect of Heat on substances
- The Kinetic Theory of matter
- Effect of impurities on melting and boiling points
- Temporary and permanent changes
- Constituents of matter
- Chemical symbols
- Chemical equations
- Revision exercise
Lesson Objectives
By the end of this chapter, the learner should be able to:
• Define mixture, element, atom, molecule, compound, melting point, boiling point and sublimation.
• Write chemical symbols of common elements and identify the constituents of compounds.
• Determine purity of a substance and use separation techniques to obtain pure substances.
• Differentiate (i) permanent and non permanent changes (ii) physical and cchemical changes.
• Explain the arrangement of particles of matter in the three physical states in terms of the kinetic theory of matter
SIMPLE CLASSIFICATION OF SUBSTANCES
The process of grouping together substances with similar properties is called classification.
Knowledge about properties can be applied to obtain pure substances from mixtures.
A. Mixtures
A mixtureis a material made up of two or more different substances which are physically combined.
In nature, matter exists often as mixtures in various combinations as 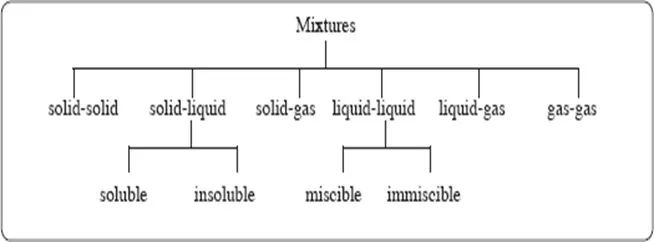 shown below.
shown below.
The method chosen to separate a given mixture depends on the physical properties of the components of the mixture.
Separation of solid-solid mixtures
In this section, other methods of separating solid-solid mixtures will be studied.
1. Using a magnet
A magnet can be used to separate a mixture such as that of iron fillings and sulphur.
Task: Place a mixture of iron fillings and sulphur on a piece of white paper and spread it out. Examine the mixture carefully and note its appearance. Hold a magnet above the mixture and observe what happens.
Questions
- Describe the colour of the mixture.
The mixture provided is grey-yellow in colour.
- What is observed when a magnet is held above the mixture?
When a magnet is held above the mixture, the grey particles are attracted leaving a yellow powder on the paper. The grey substance attracted by the magnet is iron and the yellow substance left on the paper is sulphur.
- What makes it possible for the above mixture to be separated this way?
A mixture of iron and sulphur can be separated by use of a magnet because iron is magnetic whereas sulphur is not.
- State the applications of this method of separating mixtures.
This method is applied in industries such as iron recycling, glass recycling and flour mills to remove iron particles.
2. Sublimation
Sublimation is the process where a substance changes from solid to vapour directly without forming the liquid. The solid formed when the vapour cools is known as a sublimate.
Deposition is the process by which a gas changes directly to solid (it gets deposited). It is the reverse of sublimation.
A mixture of iodine and sodium chloride can be separated by separation. To do this,
Task: Place a spatula full of the mixture in a 100ml glass beaker. Place the beaker on a tripod stand. Cover the beaker with a watch glass containing cold water.
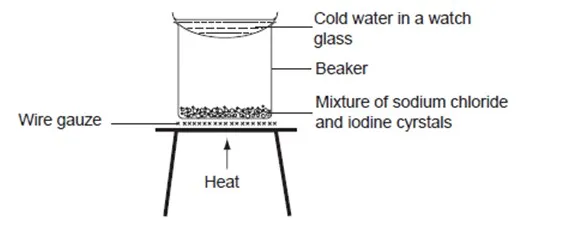
Heat the beaker gently until there is no further change. Carefully remove the watch glass from the beaker. Pour out the water from the watch glass and observe the lower surface. Examine the solid left in the beaker and record your observation.
Discussion Questions
- Describe the appearance of the mixture.
The mixture contains white and shiny-black crystals.
- What is observed inside the beaker as heating continues?
As the beaker is heated, a purple vapour is observed. On cooling, the purple vapour forms shiny black crystals on the surface of the watch glass. White crystals remain in the beaker. The shiny black crystals are iodine crystals whereas the white crystals are sodium chloride.
- What is the colour of the:
(a) solid left in the beaker? White
(b) solid formed on the surface of the watch glass?
Shiny black - Explain how the solid on the surface of the watch glass is formed.
When solid iodine is heated, it changes directly into vapour (the purple vapour seen). The vapour cools to form solid iodine on the cold watch glass.
- How is it possible to separate this mixture by sublimation?
It is possible to separate a mixture of iodine and sodium chloride because iodine sublimes while sodium chloride does not.
- Name other substances which sublime.
Other substances that sublime are anhydrous iron (III) chloride, aluminium chloride, benzoic acid and carbon (IV) oxide (dry ice).
Application of sublimation
Dry ice is used in cold boxes by ice cream vendors.
Dry ice is preferred over ordinary ice because:
- It sublimes leaving no wetness.
- It is also a better coolant compared to ordinary ice.
Solid-liquid mixtures
Substances that dissolve in a liquid are said to be soluble, while substances that do not dissolve are insoluble.
When a substance dissolves in a liquid, the substance is called a solute and the liquid is called a solvent. The resulting mixture is called a solution. When the solution is stirred, it forms a homogeneous mixture.
A Homogenous mixture is a mixture in which the solute and solvent particles are evenly distributed.
Sodium chloride, sugar, potassium nitrate, and oxalic acid crystals dissolve in water. Naphthalene, sugar and oxalic acid dissolve in propanone. Oxalic acid and sugar dissolve in both water and propanone. Naphthalene, sand and sulphur do not dissolve in water. Sodium chloride, potassium nitrate, sand and sulphur do not dissolve in propanone. Sand and sulphur did not dissolve in both water and propanone.
Separation of insoluble solid-liquid mixtures
Insoluble solid-liquid mixtures can be separated by decantation and filtration.
1. Decantation
Decantation is used to separate insoluble solid-liquid mixtures. In decantation, a solid-liquid mixture is allowed to stand such that the solid settles at the bottom. The liquid is then poured off carefully, leaving the solid behind.
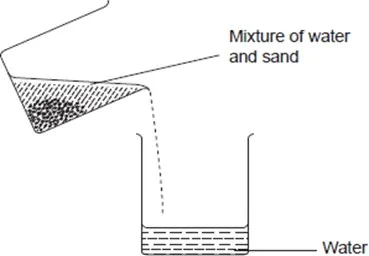
A mixture of sand and water can be separated by decantation. To do this,
Transfer 10cm3 of the mixture into 100ml beaker. Allow the mixture to settle and carefully pour the liquid into another beaker.
Discussion Questions
- What happens when the mixture is allowed to stand?
Sand is insoluble in water. When a mixture of sand and water is allowed to stand, the sand settles at the bottom. The water can then be poured off carefully.
- How clear is the water collected in the second beaker?
The water collected in the second beaker is not clear. It still contains small suspended particles.
- Why is the method not good in separating such a mixture
It is not efficient because some solids still pass into the liquid in the process of decanting. That is why the water collected in the second beaker is not clear. It still contains small suspended particles.
2. Filtration
In Filtration, an insoluble solid-liquid mixture is poured down a funnel lined with a filter paper. The solid particles are trapped and collect on the filter paper and are called the residue. The liquid passes through and is collected in the conical flask as the filtrate.
A mixture of sand and water can be separated by filtration. To do this,
Fold a filter paper into a cone shape. Place the filter paper in the funnel. Pour the mixture of sand and water carefully on the filter paper and collect the liquid in a conical flask
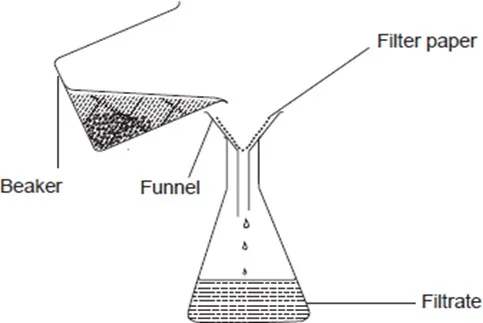
Discussion Questions
- What do you observe at the end of the experiment?
Fine sand particles are trapped and collect on the filter paper as the residue.
The filtrate (water) collected in the conical flask is clear because the filter paper does not allow solid particles to pass through.
- State an advantage of this method over decantation.
The filtrate collected in the conical flask is clear because the filter paper does not allow solid particles to pass through.
Applications of Filtration
Filtration is used on a large scale in water purification plants. Dirty water is allowed to pass through a filter bed made of layers of gravel and sand. Suspended particles are trapped by the gravel and sand while the water passes through. Domestic water filters use the same principle.
Separation of soluble solid-liquid mixtures
Soluble solid-liquid mixtures can be separated by evaporation, crystallization and simple distillation.
3. Evaporation
In evaporation, the mixture is heated to drive away the solvent in to the atmosphere. The solute remains behind.
A mixture of sodium chloride and water can be separated by evaporation. To do this;
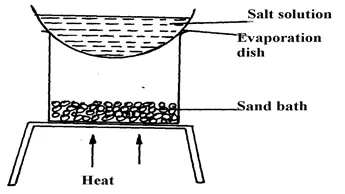
Put 10cm3 of sodium chloride solution provided into an evaporating basin. Heat the evaporating basin until crystals start forming. Transfer the evaporating basin on to a water bath and continue heating to dry the crystals. Once dry, allow the crystals to cool. A sand bath may be used in place of a water bath but it takes long to be heated up. However it retains heat much longer than the water bath.
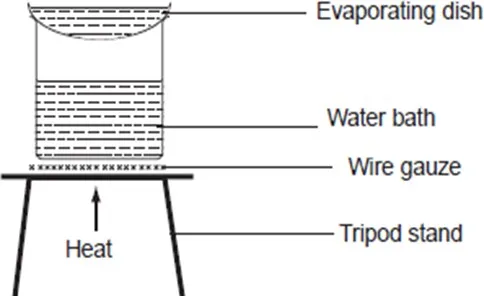
Discussion Questions
- Why was the evaporating basin containing the mixture heated?
The evaporating basin is heated to drive away water into the atmosphere.
- Why was the evaporating basin transferred to the water bath?
When crystals start forming the evaporating basin is transferred to a water bath so that the salt does not spit out of the basin as heating continues.
- State an application of this method of separation.
This process is used to obtain salt from sea water.
4. Filtration and evaporation
Both filtration and evaporation can be used to separate a mixture of two solids with one of the solids being soluble in a particular solvent whereas the other is insoluble.
A mixture of sand and sodium chloride can be separated this way. To do this;
Put a mixture of sand and sodium chloride in a beaker. Add water to it and warm while stirring. Allow the mixture to cool.
Filter the mixture using a filter paper and collect the filtrate on an evaporating basin. Evaporate the water until crystals start forming. Allow the solution to cool for more crystals to form.
Discussion Questions
- What was the residue left on the filter paper?
Sand is insoluble in water and is separated by filtration. It is collected as the residue on the filter paper.
- Name the substances contained in the filtrate.
Sodium chloride is soluble in water and is contained in the filtrate.
- Explain how sodium chloride would be recovered from the filtrate
To recover the sodium chloride, evaporation is carried out. The hot concentrated filtrate is then cooled to allow more crystals of sodium chloride to form.
- Which methods were used to separate the mixture?
Both filtration and evaporation methods were used in this experiment to separate the mixture.
5. Crystallization
Crystallisation is the process of obtaining crystals from a saturated solution.
A saturated solution is one in which no more solute can dissolve at a given temperature.
This method can be used to separate most soluble substances from their solutions.
Crystals of various salts such as copper (II) sulphate can be obtained by crystallisation. To do this;
Put 10cm3 of concentrated copper (II) sulphate solution into an evaporating basin. Arrange the apparatus as shown below.
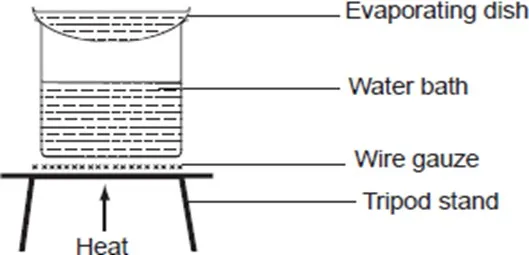
Using a water bath, heat the solution to evaporate excess water. As heating continues, dip a glass rod into the solution regularly and allow it to cool in the air. When crystals start forming on the glass rod, remove the evaporating basin from the water bath and allow it to cool.
Discussion Questions
- Why is the glass rod dipped in the solution while heating continues?
The glass rod is dipped into the solution to find out whether the solution can form crystals on cooling. When crystals form on the glass rod, this is an indication that the solution is ready to form crystals. At this point the solution is said to be saturated.
2. What observations are made when the solution cools?
Blue Crystals of copper(II) sulphate form.
Application of crystallisation
1. Extraction of salt from salty water e.g Lake Magadi and Ngomeni in Malindi.
2. Extraction of sugar from sugar cane.
3. Extraction of medicinal substances from plants.
6. Simple distillation
Distillation is the process of evaporating a liquid from a solution and condensing the vapour produced back into liquid.
The liquid that is obtained after condensation is called the distillate.
Simple distillation can be used to obtain a solvent from a solution. In this method, both the solvent and the solute can be collected.
Water can be obtained from a solution of sodium chloride using simple distillation. To do this,
Place about 20cm3 of sodium chloride solution in a boiling tube or a round-bottomed flask. Arrange the apparatus as shown below.
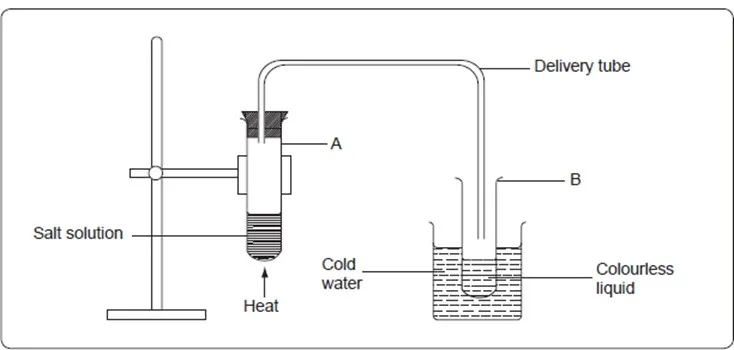
Heat the solution until all the solvent has evaporated. Record your observation.
Place about 20cm3 of the sodium chloride solution on a watch glass. Place the watch glass on a water bath. Heat the sodium chloride solution to dryness. Remove the watch glass from the water bath and allow it to cool. Record your observations.
Place the liquid collected in tube B on another watch glass. Place the watch glass on the water bath. Heat the liquid to dryness. Allow the watch glass to cool. Record your observation.
Discussion Questions
- What is observed in the delivery tube as the solution boils?
The steam passes through the delivery tube where some of it condenses on the cooler parts of the delivery tube to a colourless liquid. The steam is cooled and condenses to liquid water in test state tube B. The liquid collected in test tube B is called the distillate.
- What is the purpose of cold water in the beaker?
The cold water in the beaker acts as a coolant.
3. What is observed in test state tube A after all the solvent has evaporated?
A white solid remains in tube A. The white solid is sodium chloride. This method of separating a solute from a solvent is called simple distillation.
4. What is observed on the watch glass containing:
(i) sodium chloride solution?
When sodium chloride solution is placed on a watch glass and evaporated to dryness, a white residue is left behind.
(ii) liquid from test state tube B when heated to dryness? Explain.
When the distillate is placed on a watch glass and evaporated, no residue remains. This shows that the distillate no longer contains dissolved sodium chloride.
To improve the condensation process, a Liebig condenser is used as shown below.
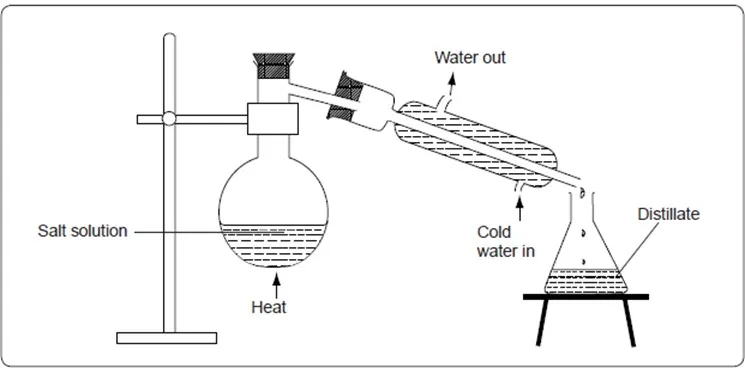
Simple distillation with Liebig condenser.
Liquid-liquid mixtures
There are two types of liquid-liquid mixtures namely immiscible and miscible liquids.
Immiscible liquids do not mix but form distinct layers. Examples of such mixtures are vegetable oil and water, kerosene and water.
Miscible liquids mix to form a homogenous mixture. Examples are water and ethanol, water and milk.
Separating Immiscible Liquids
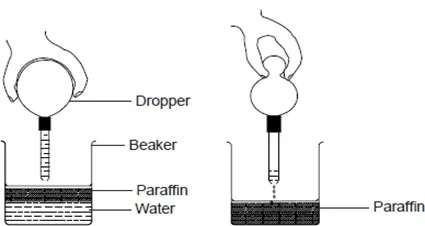
Immiscible liquids can be separated using a separating funnel, dropper or by decantation. A burette may also be used.
1. Using a separating funnel
A mixture of kerosene and water can be separated using a separating funnel. To do this,
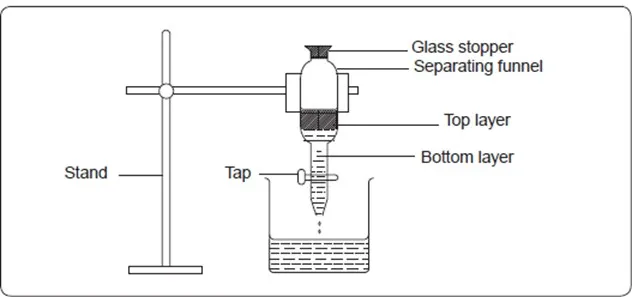
Put 10cm3 of coloured water in a conical flask. Add 10cm3 of kerosene and shake well. Transfer the mixture into a separating funnel. Allow the mixture to stand for a while and observe. Remove the stopper. Open the tap of the separating funnel and allow the bottom layer to flow into a beaker. Discard the interphase (the boundary between water and kerosene) leaving the top layer in the separating funnel.
Discussion Questions
- Which layer is at the top in the separating funnel? Explain.
Kerosene. Kerosene floats on water because it is lighter.
- Why is the water coloured?
The water is coloured to distinguish it from kerosene.
- Explain why the interphase was discarded.
The interphase contains both water and Kerosene. It is discarded because it is not easy to separate the two liquids at the interphase.
- What property of the mixture makes it possible to be separated this way?
A mixture of water and kerosene is immiscible. The two liquids also have different densities.
- Which other method could be used to separate the two layers?
Decantation can also be used to separate immiscible liquids but it is not efficient.

A dropper can also be used to separate immiscible liquids by sucking the upper layer.

The dropper is used to suck one layer transferring it to another beaker repeatedly. This method too is not accurate.
Separating Miscible Liquids
Miscible liquids can be separated using fractional distillation. The two liquids must be having different but close boiling points.
1. Separating a mixture of water and ethanol using fractional distillation
Place 20cm3 of water in a round-bottomed flask. Add 20cm3 of ethanol and shake the mixture. Set up the apparatus as shown below.
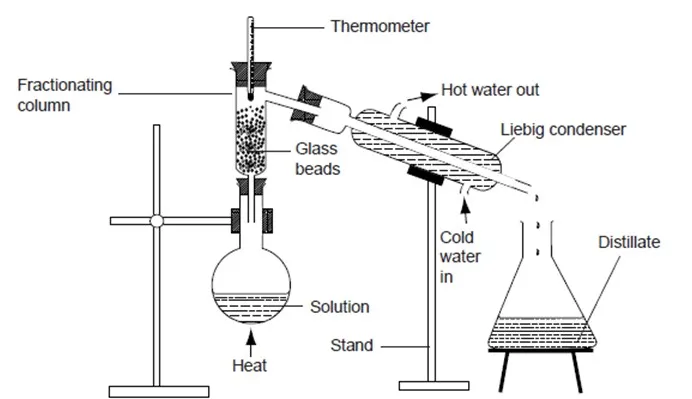
Heat the mixture gently and note the temperature changes. Collect the distillate in the conical flask. Transfer a little distillate on a watch glass and light it. Record your observations.
Discussion Questions
- Why is it possible to separate ethanol from water?
Water and ethanol are miscible liquids. They are separated because they have different but close boiling points. Pure ethanol boils at 78.2°C while pure water boils at 100°C at sea level.
- Explain the role played by the fractionating column.
The purpose of the fractionating column is to allow water vapour to condense into liquid and flow back into the flask before the boiling point of water is reached.
- What is the role of the thermometer?
To indicate when the boiling point of the medium being distilled has been reached.
- Which substance is collected as the first fraction? How is water collected?
When the mixture is heated, ethanol boils off first at about 78°C, and is collected as the first fraction of the mixture.
The temperature remains fairly constant until the ethanol distils off. At this stage the temperature starts rising and the distillate collected thereafter is mainly water as a second fraction.
- State the role of the glass beads in the fractionating column.
The glass beads increase the surface area for condensation.
- What is the role of the Liebig’s condenser?
A Liebig condenser uses the counter flow principle to cool the vapour efficiently.
- What would happen if the inlet and outlet were exchanged in the Liebig’s condenser?
If the inlet and outlet were exchanged in the Liebig condenser, condensation would still occur but less efficiently.
Applications of fractional distillation
1. Distillation of:
(a) crude oil to obtain fractions such as diesel, petroleum, cooking gas. (Kenya Petroleum Refinery in Changamwe, Mombasa),
(b) recycling of used oil (at Athi River and Kikuyu town).
2. Liquid air in the manufacture of nitrogen and oxygen. (British Oxygen Company, BOC).
2. Solvent extraction
Solvent extraction, also known as liquid-liquid extraction is a process that allows the separation of two or more components due to their unequal solubilities. The solvent, also known as an extractant is chosen to selectively extract a certain component from a mixture.
Groundnuts, cashew nuts and coconuts contain oil which is useful. The oil can be extracted by solvent extraction using a suitable solvent such as propanone.
How to extract oil from groundnuts
Crush about 20 groundnut seeds in a mortar using a pestle. Continue crushing the nuts while adding propanone a little at a time. Decant the resulting solution into an evaporating basin. Leave the solution in the sun for sometime.
Discussion Questions
- Why are the nuts first crushed?
The nuts are first crushed to increase the surface area in contact with the solvent.
- What is the role of propanone in the experiment?
Propanone is used as a solvent because it dissolves oil
- Explain why water is not used as a solvent in this experiment.
Water which is a common solvent cannot be used in this extraction because it will not dissolve oil.
- Why is the solution left in the sun?
Once the oil has dissolved in the propanone the solution is left in the sun for the solvent to evaporate. The oil having a higher boiling point than the solvent is left in the evaporating dish.
- How can the oil obtained this way be made more pure?
Oil obtained this way can be made more pure by washing the product in water and separated from the water using a separating funnel.
- How can the presence of the oil be tested?
Oils leave a translucent patch on paper. This can be used as a simple test for their presence.
Applications of solvent extraction
1. Extraction of:
(i) oil from nuts and seeds.
(ii) natural dyes from plants.
(iii) some herbal medicines from plants
(iv) caffeine from tea and coffee
Review Exercise 1.
- Study the information below and answer the following questions. A mixture contains three solids A, B, and C. The solubility of these solids in different liquids is as shown below:-
Solid | Water | Alcohol | Ether |
A | Soluble | Insoluble | Insoluble |
B | Insoluble | Soluble | Very soluble |
C | Soluble | Soluble | Insoluble |
Explain how you will obtain sample C from the mixture
2. State and explain the observations made when iodine crystals is heated in a boiling tube?
3. Study the information in the table below and answer the questions.
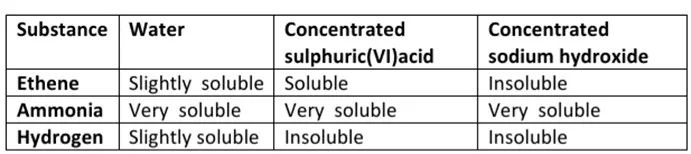
A mixture contains ethene, Hydrogen and ammonia gases. Explain how a sample of hydrogen gas can be obtained from this mixture
4. Study the information below and answer the questions that follow:
Solid | Cold water | Hot water |
R | Soluble | Soluble |
V | Insoluble | Insoluble |
S | Insoluble | Insoluble |
Describe how the mixture of solid R, S, and V can be separated
of 23oC to 27oC.
(i) What evidence is then to support the statement that cooking oil is a mixture?
(ii)Name another experimental technique that could be used to confirm your answer
in part (i) above
7. A sample of copper turnings was found to be contaminated with copper (II) oxide. Describe how a sample of copper metal can be separated from the mixture.
8. A form 1 student carried out the separation as shown in the set-up below:-
(i) Identify the method above………………………………………………………………………
(ii) Give one of its disadvantages
(iii) Name a mixture which can be separated by the set-up above
9. The set-up below was used to separate a mixture:-
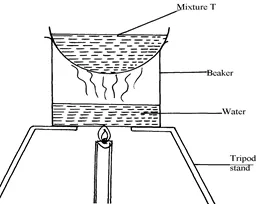
(a) Name the apparatus missing in the set-up
(b) Give one example of mixture T
(c) What is the name of this method of separation
10. A student left some crushed fruit mixture with water for some days. He found the mixture had fermented. He concluded that the mixture was contaminated with water and ethanol with boiling point of 100oC and 78oC respectively. The set-up of apparatus below are used to separate the mixture.
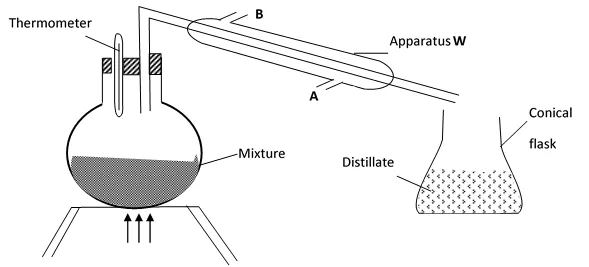
(i) Name the piece of apparatus labelled W
(ii) What is the purpose of the thermometer in the set-up?
iii) At which end of the apparatus W should tap water be connected?……………………………
(iv) Which liquid was collected as the first distillate? Explain
(v) What is the name given to the above method of separating mixture?
(vi) State two applications of the above method of separating mixtures
(vi) What properties of the mixture makes it possible for the component to be separated by the above methods?
11. The setup below was used to separate two miscible liquids Q and T (Boling points; Q =98° C, T=78°C)
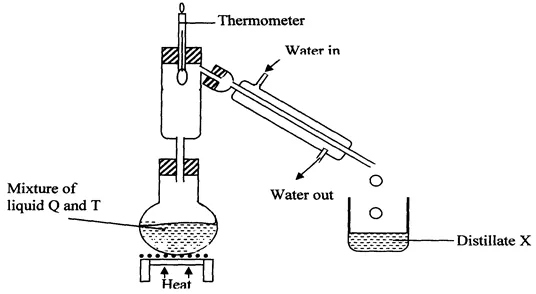
(a) Identify the mistakes in the setup above
(b) Identify Distillate X
Separation of Coloured substances
Naturally occurring substances may contain several pigments (colours). For example green grass has several pigments. The colours can be separated by chromatography.
Chromatography
Chromatography is derived from the terms
- Chroma which means colour and
- chromatology which means study of colours
Chromatography is a method of separating coloured substances. Each coloured substance has a different solubility in a solvent and a different extent of adsorption on the adsorbent material.
Adsorption involves the binding of molecules or particles of a gas or liquid to the surface of a solid.
A chromatogram
is a final representation of the separated pigments.
The furthest point where the solvent reaches on the adsorbent material is called the solvent front.
Using chromatography to separate a mixture of pigments
Crush some green leaves or grass in a mortar using a pestle. Add propanone as you continue crushing the leaves.
Decant the extract into a beaker. Place a filter paper on top of an empty beaker. Using a dropper place one drop of the extract at the centre of the filter paper and allow it to spread as far as possible. Add a second and third drop at the same spot. Each time allow the extract to spread as far as possible.
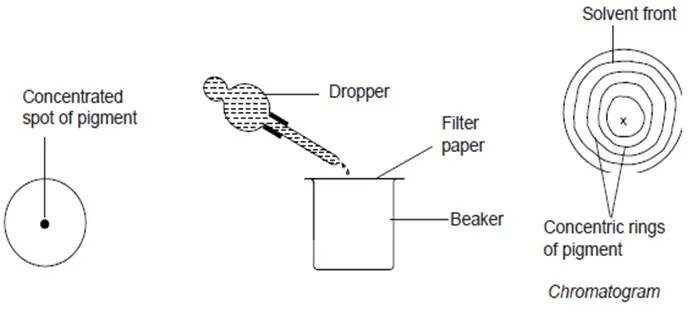
Once the spot of the extract has stopped spreading, add the solvent drop-wise each time allowing the solvent to spread. Continue adding the solvent until it spreads out close to the edge of the filter paper. Draw the diagram of the filter paper showing the results obtained.
Discussion Questions
- What is the role of propanone?
The colouring matter in green leaves is composed of different substances. Propanone is used to dissolve the colouring matter.
- What is observed at the end of the experiment?How many pigments are there in green leaves extract?
The coloured matter in leaves separates into two distinct pigments; a green colour due to a substance called chlorophyll and a yellow colour due to xanthophylls. The dry filter paper showing the separated pigments is called chromatogram. Xanthophyll is more soluble and less adsorbed.
- Explain how the pigments separate?
Each coloured substance has a different solubility in propanone and a different extent of adsorption on the filter paper (adsorbent material). As the propanone spreads, the pigments which are more soluble and less adsorbed are carried furthest while the less soluble and more adsorbed are left behind; as a result separation takes place.
Using chromatography to determine the presence of a substance in a mixture
Chromatography can be used to determine the presence of a substance in a mixture by comparing it with a pure substance. The suspect mixture is placed on an adsorbent medium alongside the pure substance on the same baseline.
The paper strip is then placed in a beaker containing a solvent.
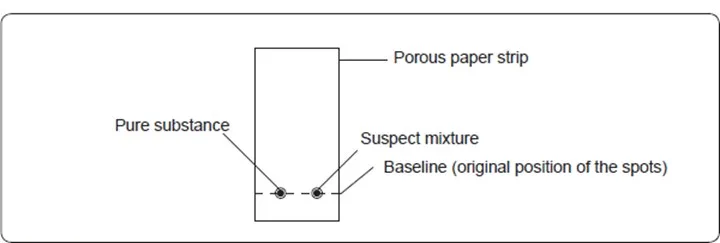
The solvent is allowed to ascend to the top (solvent front) and the paper is then removed and allowed to dry. The position of the spots from the mixture and the pure substance are noted and compared.
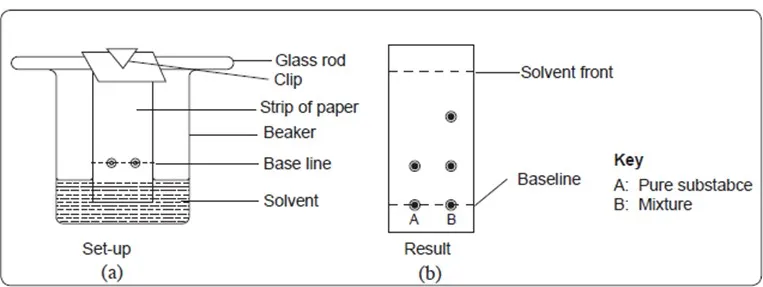
If any of the spots in the mixture moves the same distance as the spot in the pure substance, then the mixture contains the pure substance as one of the components.
This procedure is known as ascending paper chromatography.
Applications of chromatography
1. In sports, chromatography, is used to identify banned substances, for example steroids in urine or in blood samples.
2. In the pharmaceutical industry, to test purity of drugs.
3. In food industry, to identify contaminants in food and drinks.
4. In the cosmetics industry, to identify harmful substances.
Review Exercise 2
- The following diagram shows a paper chromatogram of substances A, B, C, and D which are coloured
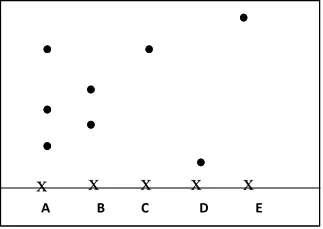
(a) Indicate the solvent front on the chromatogram
(b) Which substance is pure? ………………………………………..
(c) Substance E is a mixture of C and D. Indicate its chromatogram in the diagram
2. The chromatography below shows the constituents of a flower extract using an organic solvent:-
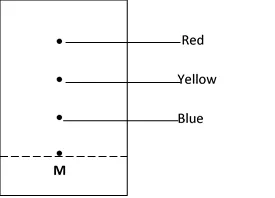
(a) (i) Name a possible organic solvent you can use for this experiment
(ii) State one property that makes the red pigment to move the furthest distance from M
(iii) Describe how one could get a sample of yellow pigment
(iv) On the diagram indicate solvent front
(b) Describe how Aluminium chloride can be separated from a mixture of aluminium chloride and sodium chloride
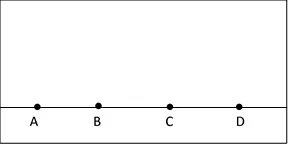
After development A, B, and C were found to have moved 8cm, 3cm and 6cm respectively.D had separated into two spots which had moved 6cm and 8cm
On the diagram above;
I. Label the baseline (origin)
II. Show the positions of all the spots after development
ii) Identify the substances present in mixture D
b) Describe how solid ammonium chloride can be separated from a solid mixture of
ammonium chloride and anhydrous calcium chloride
c) The table below shows liquids that are miscible and those that are immiscible
Liquid | L3 | L4 |
L1 | Miscible | Miscible |
L2 | Miscible | Immiscible |
Use the information given in the table to answer that questions that follow;
i) Name the method that can be used to separate L1 and L2 from a mixture of the two
ii) Describe how a mixture of L2 and L4 can be separated
4. The diagram below shows chromatograms of blood samples obtained from three athletes.
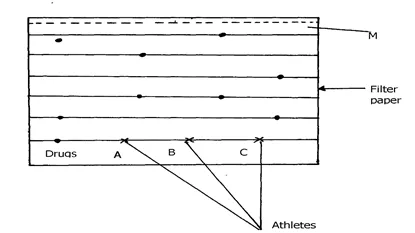 One athlete used illegal drug to improve performance in competition.
One athlete used illegal drug to improve performance in competition.





(a) Name the line marked M………………………………………………….
(b)Identify the athlete who used illegal drug ……………… ……………………….
5. (a) What is chromatography?
b) Give two applications of chromatography
6. The paper chromatography of a plant extract gave the following results:
Solvent | Number of spots |
X | 6 |
Y | 2 |
Z | 3 |
(a) Which is the most suitable solvent for purifying the extract? Explain
(b) Ball pen cannot be used to mark solvent front in the above chromatography. Explain
B. Effect of Heat on Substances
States of Matter
Matter is anything that has mass and occupies space. Matter exists in three physical states namely solid, liquid and gas. A substance can exist in any of the three states depending on the prevailing temperature.
Heating and cooling curves of solid naphthalene
Place a spatulaful of naphthalene in a boiling tube. Note the temperature of the naphthalene. Place the boiling tube in a beaker of water as shown below and heat the beaker. Record the temperature of the naphthalene every half a minute throughout the experiment until all the solid has melted. Continue heating for about two more minutes. Record the temperature readings in a table and plot a graph of temperature against time.
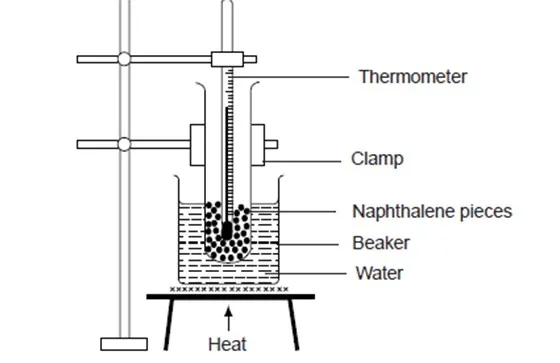
Discussion Questions
- What happens to naphthalene when it is heated? Explain.
When naphthalene is heated, its temperature rises steadily until it starts to melt. At this point, the temperature remains constant until all the naphthalene has melted then the temperature starts rising again.
- Why is heating done in a water bath?
Heating in a water bath does not allow the boiling point of naphthalene (218°C) to be attained and therefore protects the thermometer from breaking.
- Is there a temperature change as naphthalene is melting?
No. Temperature remains constant when naphthalene is melting.
- Explain what happens as heating of naphthalene continues. How does heating cause melting?
A sketch of the graph of temperature against time obtained when naphthalene is heated to melting is shown below. Such a graph is called a heating curve.
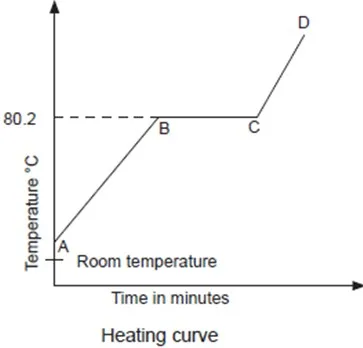
Region AB:The temperature increases steadily as the naphthalene absorbs heat energy. The heat absorbed increases the kinetic energy of the particles and they vibrate more vigorously.
Region BC:The temperature remains constant until all the naphthalene melts. Here, the heat supplied is used to weaken the forces of attraction holding the particles of naphthalene together. The particles therefore move far apart. As a result, the naphthalene changes its state from solid to liquid.
Region CD:Temperature rises steadily as the liquid naphthalene absorbs heat energy. The heat supplied increases further the kinetic energy of the particles causing them to move fast.
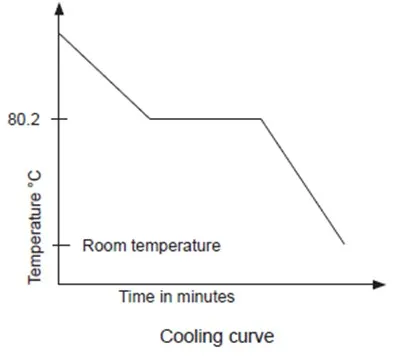
5. Explain how the change observed in the experiment can be reversed.
If liquid naphthalene is allowed to cool, the reverse can be obtained. It is then referred to as cooling curve.
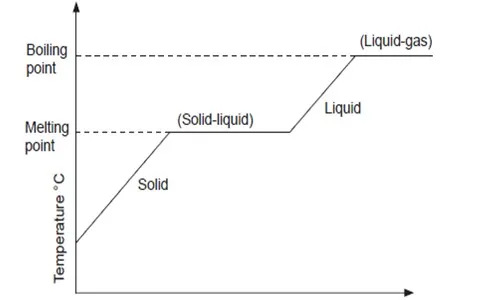
The figure below represents a heating curve of a solid which is heated from room temperature to boiling point.
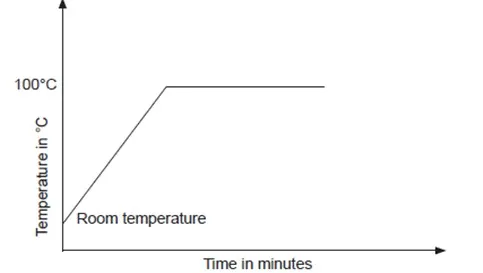
Heating curve for a liquid: pure water
When water is heated, its temperature steadily increases as molecules absorb heat energy, which increases their kinetic energy. The temperature of the water continues to rise until the water starts to boil.
The temperature of the water remains constant as it boils. This is because the heat energy supplied is used to break the forces of attraction holding the particles together. As a result some particles break free and the water changes from liquid to gaseous state.
The graph of temperature against time obtained when water is heated to boiling is given below.
Change of state from solid to liquid and liquid to gas can be reversed by cooling. On cooling, the gas condenses into liquid and finally the liquid freezes into solid. These changes are shown in the flow diagram below.
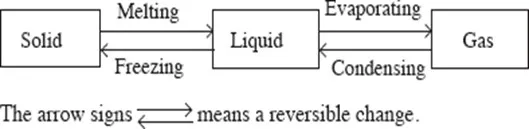
The use of the thermometer enables us to observe that changes in the states of matter are not accompanied by temperature changes. Some forces of attraction hold together the particles which make up matter. Heating provides the energy required to overcome these forces.
Some substances such as iodine do not undergo the above changes because they sublime.
The explanation for the behaviour of matter in terms of the movement of particles is provided by the kinetic theory of matter.
C. The Kinetic Theory of Matter
According to the theory, matter is made up of particles which are in a continuous state of motion. The kinetic theory forms the basis of the theoretical model of matter.
The figure below shows the model in which circles represent small particles of matter.
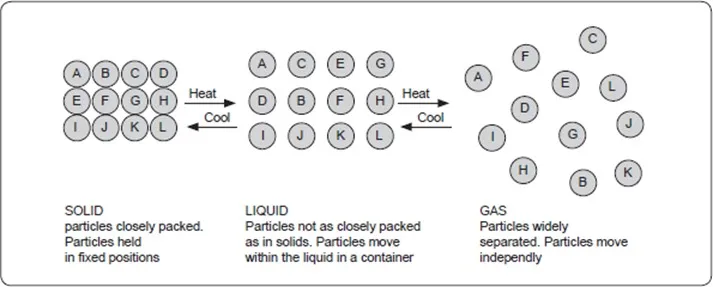
Solid state
In the solid state, the particles are closely packed together and can only vibrate within fixed positions. They do not move from one point to another because there are forces that hold them in these positions. When a solid is heated, the kinetic energy of the particles increases and they begin to vibrate more vigorously. At a certain temperature which is fixed for particular substance, the forces holding the particles are weakened enough to allow the particles to change position as a result the solid changes into a liquid. This temperature is known as the melting point.
Liquid state
In the liquid state, particles are not as close together as they are in the solid state. They can move from one position to another within the liquid.
This explains why a liquid has no definite shape and will take the shape of the container. However, a liquid has a definite volume. The particles exert some attraction on one another and these forces of attraction make them to stay together.
When a liquid is heated, the particles move more rapidly as the forces of attraction are further weakened. The weakening continues until the particles gain enough energy to overcome the forces between them. At this point, the liquid boils as particles break free and enter the gaseous phase/state. The constant temperature at which a pure liquid boils is called the boiling point and is fixed for a particular substance. The temperature at which a liquid boils depends on the external atmospheric pressure.
Gaseous state
In the gaseous state the particles are far apart and free to move randomly in all directions. This is why a gas does not have definite shape or volume but occupies the whole space within a container.
When a gas is cooled the particles lose kinetic energy and hence slow down. As they slow down they easily attract their neighbouring particles and move close to form a liquid. This process is called condensation.
Condensation occurs at the same temperature as evaporation.
On further cooling of the liquid, the kinetic energy of the particles continues to decrease and the particles take up fixed positions as the liquid solidifies. This process is called freezing. Freezing takes place at the same temperature as melting.
Effect of Impurities on Melting and Boiling Points of Substances
a) Effect of Impurities on Melting point
Impurities lower the melting point of substances. Impure substance do not have sharp melting points.
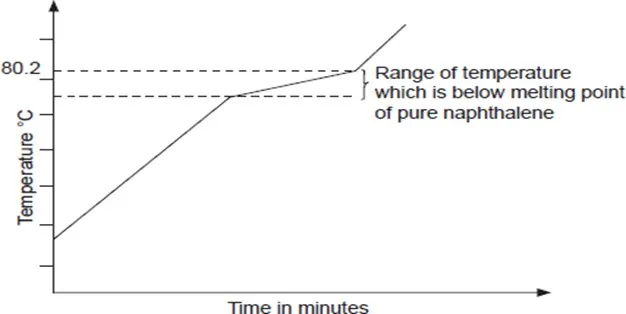
For example, the melting point of pure naphthalene is 80.2°C. If camphor or another substance is added as an impurity, the naphthalene melts over a temperature range that is lower than the melting point of pure naphthalene. Impure naphthalene does not have a sharp melting point. Impurities therefore lower the melting point of substances.
Applications of Lowering Melting Point by Impurities
The lowering of melting point by impurities is applied in:
1. Clearing of ice from roads in temperate countries by spreading common salt on the ice.
2. Extraction of metals from their molten compounds. For example, calcium chloride is added to rock salt during the extraction of sodium from sodium chloride.
(b) Effect of impurities on the boiling points of substances
Impurities raise the boiling points of substances. Impure substances do not have sharp boiling points.
For example, the boiling point of pure water is 100°C at sea level. However, impure water starts to boil at a temperature above 100°C. The temperature continues to rise as the impure water boils. Thus the impure water boils over a range of temperature. Impurities raise the boiling point of a liquid. The heating curves for pure and impure water are shown in (a) and (b).
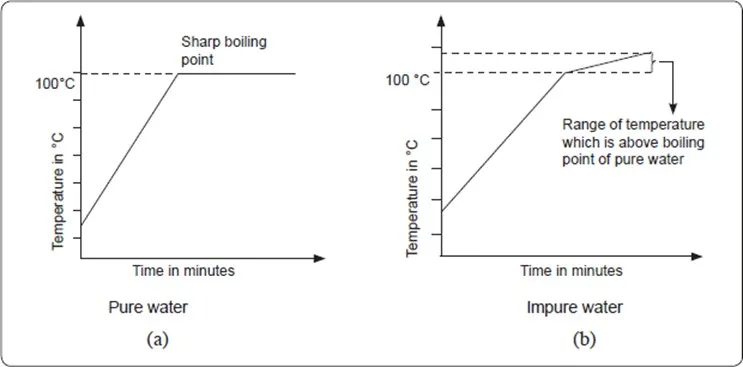
Criteria of Purity
Pure substances melt and boil at constant temperatures
that are specific for a particular substance. Melting and boiling points are therefore used for determining purity of substances.
Review Exercise 3
- Substance Q has a melting point of 15oC and boiling point of 70oC.
(a) On the same axes, draw the melting point and boiling point graph for Q and the room Temperature.
(b) State the physical state of substance Q at room temperature
2. The diagram below shows the heating curve of a pure substance. Study it and answer the questions that follow:
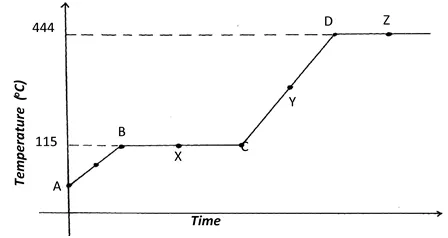
(a) What physical changes are taking place at points X and Z?
(b)Explain what happens to the melting point of sodium chloride added to this substance
3. What is meant by melting point and boiling point of a substance?
4. State two criteria for determining the purity of a substance
D. Temporary and permanent changes
Substances undergo various changes when subjected to different conditions of temperature.
Temporary changes
A temporary change may be physical or chemical. In a temporary physical change, no new substance is formed. In a temporary chemical change new substances are formed. However the reactions are reversible if the conditions are reversed.
I) Temporary physical change
A change which can easily be reversed and in which no new substance is formed is called a temporary physical change.
Characteristics of temporary physical changes.
(a) They are easily reversible.
(b) No new substance is formed.
(c) The mass of the substance does not change.
(d) They are not accompanied by net heat change.
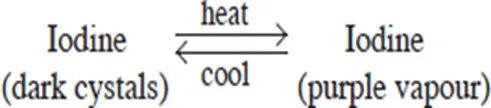
Examples of temporary physical changes are heating zinc oxide, wax and iodine which does not result in the formation of a new substance. Cooling reverses the changes these substances undergo.
- When zinc oxide is heated, its colour changes from white to yellow. On cooling, the yellow solid turns white. This change can be represented as:

- Wax melts on heating. When cooled, liquid wax changes back to solid.

- When iodine is heated, the shiny black solid turns to a purple vapour. When cooled, the purple vapour changes back to the shiny black solid.
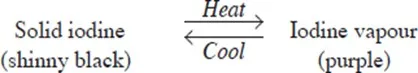
II) Temporary chemical change
A temporary chemical change is a change which can be reversed but a new substance is formed.
Characteristics of temporary chemical change
(a) A new substance is formed.
(b) Heat energy is evolved or absorbed.
(c) There is change in mass.
(d) The change can be reversed.
Heating hydrated copper (II) sulphate or hydrated cobalt (II) chloride results into a temporary chemical change.
- A hydrated substance is that which contains water of crystallization.
When hydrated copper(II) sulphate is heated, it decomposes to produce white copper (II) sulphate powder and water. The white copper (II) sulphate powder does not contain water of crystallisation and is said to be anhydrous.
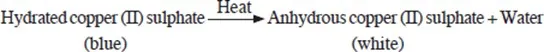
The white anhydrous copper (II) sulphate does not regain the original blue colour on cooling.
Similarly pink cobalt (II) chloride decomposes to form blue anhydrous cobalt (II) chloride and water vapour.

The blue cobalt (II) chloride does not regain the pink colour on cooling.
- The decomposition of a substance when it is heated is referred to as thermal dissociation.
Heat is evolved when a little water is added to the anhydrous copper sulphate or the blue anhydrous cobalt (II) chloride. The anhydrous substances become hydrated and regain their
original colour.
Permanent chemical change
A permanent chemical change is a change in which a new substance is formed and the change cannot be reversed.
Characteristics of permanent chemical changes
(a) New substances are formed.
(b) The change is irreversible.
(c) The change is accompanied by change in mass.
(d) Heat energy is released or absorbed.
The decomposition of copper (II) nitrate and potassium manganate (VII) are examples of permanent chemical changes.
When copper (II) nitrate is heated it decomposes to form a black solid and a mixture of gases. The black solid is copper (II) oxide. The mixture of gases consists of a red brown gas and another gas which relights a glowing splint. The red brown gas is nitrogen (IV) oxide while the gas which relights the glowing splint is oxygen.

The mass of copper (II) oxide is found to be less than that of copper (II) nitrate because the gaseous products escaped into the atmosphere.
Also, Potassium manganate (VII) decomposes to form a black-green solid and a colourless gas, which relights a glowing splint. The black-green solid is a mixture of black manganese (IV) oxide and green potassium manganate (VI). The black-green solid weighs less than the original potassium manganate (VII).

Review Exercise 4
- The apparatus below were used by a student to study the effect of heat on hydrated
copper II sulphate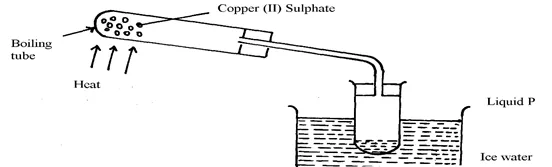
(a) What is the role of the ice cold water ……………
(b) Name liquid P…………………………………………………………
(c) What observation is made in the boiling tube
2. Classify the following processes as chemical changes or physical changes
Process
physical or chemical
Neutralization ………………………………………
Sublimation ………………………………………
Fractional distillation ………………………………………..
Displacement reaction …………………………………………
b) Pellets of sodium hydrogen and anhydrous Copper (II) sulphate were put in separate Petri- dishes and left in the open for two hours. Explain the observation in each Petri-dish
E. Constituents of Matter
Pure substances can be classified as elements or compounds.
Elements
Elements are pure substances which can not be split into simpler substances by chemical means.
Examples of elements include oxygen, hydrogen, copper, sulphur, carbon and iron. There are over a hundred known elements.
Elements are made up of atoms.
The atom is defined as the smallest particle of an element, which can take part in a chemical change. Atoms of the same element are similar.
The atoms of some elements can not exist independently but join together to form small groups of atoms. These discreet particles are called molecules.
A molecule is defined as the smallest particle of an element or compound, which can exist separately.
Compounds
When two or more elements combine chemically they form a compound.
A compound is a pure substance made up of two or more elements chemically combined.
The compound formed when iron and sulphur chemically combine is called iron (II) sulphide.
In compounds, names ending in –ide means the compound is composed of two elements only. e.g:
Sodium Chloride is made up of Sodium and Chlorine.
Iron (II) Sulphide is made up of Iron and Chlorine.
Calcium Nitride is made up of Calcium and nitrogen.
Calcium carbide is made up of Calcium and Carbon.
Sodium hydride is made up of Sodium and hydrogen.
An exception to this are the hydroxides.
Names ending in –ate means the compound is composed of three elements one of which is oxygen. eg:
Sodium Sulphate is made up of sodium, sulphur and oxygen.
Sodium Carbonate is made up of sodium, carbon and oxygen.
Potassium chlorate is made up of potassium, chlorine and oxygen.
Calcium nitrate is made up of calcium, nitrogen and oxygen.
Exceptions to this rule are the hydrogen carbonates and hydrogensulphates.
Names ending in
–ite means the compound is made up of three elements one of which is oxygen. However, the amount of oxygen is less than in those compounds whose names end in –ate e.g
Sodium Sulphite is made up of sodium, sulphur and oxygen.
Calcium Sulphite is made up of calcium, sulphur and oxygen.
Chemical Symbols
In Chemistry, elements are represented by letters. The letters are referred to as chemical symbols. A chemical symbol of an element is usually the first letter or the first and another letter of the element’s English or Latin name.
The first letter of a chemical symbol must always be a capital letter while the second letter is always a small letter.
Symbols derived from first letter of name of element
Element | Symbol |
Carbon | C |
Nitrogen | N |
Oxygen | O |
Hydrogen | H |
Symbols of several elements may begin with the same letter. It is therefore necessary to represent some of these elements with two letters as shown in the following table. The second letter in a chemical symbol is always a small letter.
Symbols derived from first and second letter of name of element
Elements | Symbol |
Calcium | Ca |
Cobalt | Co |
Chlorine | Cl |
Magnesium | Mg |
Manganese | Mn |
In some cases, the symbol of the element is derived from the element’s Latin name as shown in the table below.
Symbols derived from latin name of element
Element | Latin name | Symbol |
Potassium | Kalium | K |
Sodium | Natrium | Na |
Silver | Argentum | Ag |
Gold | Aurum | Au |
Iron | Ferrum | Fe |
Lead | Plumbum | Pb |
Mercury | Hydragyrum | Hg |
Copper | Cuprum | Cu |
F. Chemical Equations
Iron and sulphur combine chemically to form iron (II) sulphide. In this process iron and sulphur are referred to as the reactants whereas the iron (II) sulphide is referred to as the product.
Chemical combination of elements is known as a reaction.

When a reaction is represented as shown, the representation is called a word equation. In a chemical equation reactants are written on the left hand side of the arrow sign  while the products are written on the right hand side.
while the products are written on the right hand side.
The forward arrow sign  is used where the reactions are permanent and proceed only in one direction.
is used where the reactions are permanent and proceed only in one direction.
Two arrow signs in opposite directions  are used where the reactions are reversible. This means the reaction can proceed in either direction.
are used where the reactions are reversible. This means the reaction can proceed in either direction.
NOTE:
The plus sign on the reactants side means “react with.”
The plus sign on the product side means “and”
The arrow sign between the reactants and the products means to “yield”.
1. Iron reacting with sulphur

2. Sulphur reacting with oxygen

3. Carbon reacting with oxygen

4. Sublimation of Iodine

Revision Exercise On Simple Classification of Substances
1. Explain the differences between solid, liquid and gaseous states using the theoretical model of matter in terms of the kinetic theory.
2. The graph below shows the shape of the curve obtained by a student when solid X was heated to boiling.
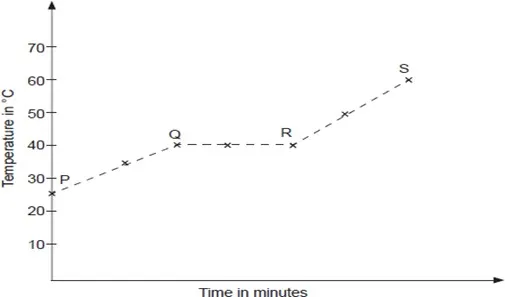
(a) (i) Determine the melting point of solid X.
(ii) State and explain what portions PQ, QR and RS represent.
(b) If candle wax was used in this experiment, the portions QR would not be horizontal. What does this tell us about candle wax.
3. Name four examples of;
(a) mixtures
(b) compounds
(c) elements
4. Name the elements present in the following compounds.
(a) Sodium bromide
(b) Zinc sulphide
(c) Lead oxide
(d) Magnesium nitride
(e) Potassium iodide
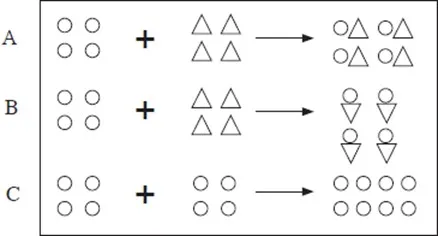
5. Which of the following best shows two elements combining to form a compound? Explain.
6. Name a solvent that can be used for the following:
(a) removing ink from clothes
(b) cleaning greasy hands
7. How can one separate and collect the solvent from a salt solution?
8. Explain how Elianto oil is obtained from maize seeds.
9. Give two examples of:
(i) temporary physical change.
(ii) temporary chemical change.
(iii) permanent change.
10. List three differences between temporary and permanent changes.
11. Spots of pure pigments A and B, and a mixture Z were placed on a filter paper and allowed to dry. The paper was then dipped in a solvent. The results obtained were as on the paper chromatogram.
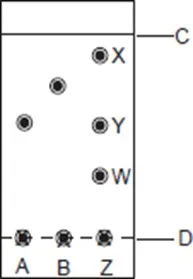
(a) Which is the:
(i) base line?
(ii) solvent front?
(b) Which of the pure pigments was a component of Z? Explain.
(c) (i) Name a solvent that is used in paper chromatography.
(ii) Why is water not a suitable solvent in paper chromatography?
12. Write a word equation for the reaction between:
(i) carbon and oxygen
(ii) sodium and sulphur
(iii) copper and chlorine
13. Name the process which takes place when:
(a) Solid Carbon (Iv) Oxide (dry ice) changes directly into gas
(b) A red litmus paper turns white when dropped into chlorine water
(c) Propene gas molecules are converted into a giant molecule



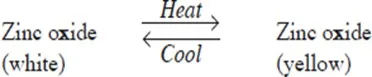
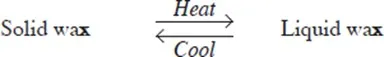
Comments are closed.